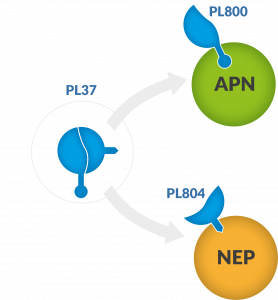
PL37 is a Dual ENKephalinase Inhibitor (DENKI), a small molecule with two active metabolites (PL800 & PL804), which acts by inhibiting the enkephalin-degrading enzymes APN (aminopeptidase N) and NEP (neprilysin) respectively, thereby increasing the local concentration of enkephalins triggering a local and sustainable pain relief.
PL37 has completed an oral phase IIa study evaluating the safety, tolerability and efficacy of 200 mg t.i.d. of PL37 in patients with peripheral neuropathic pain of diabetic origin unsatisfactorily treated with either pregabalin or gabapentin.
An analysis of the pregabalin subgroup showed that PL37 met the study’s primary endpoint in this patient population with a clear difference of 1.21 between PL37 and placebo. The pain scores of patients treated with PL37 kept improving throughout the study while the safety and tolerability of PL37 was good.
PL37 pharmacokinetics has shown that its target enzyme inhibition is effective and short lived, making it an ideal development candidate for intravenous or acute oral administration.
Oral PL37 for the acute treatment of episodic migraine
Migraine is a frequent and debilitating painful disease, affecting more than 12% of the general population. NSAIDs and triptans are currently used for the acute treatment of migraine attacks. However, these treatments are not effective or well-tolerated in all patients. Despite the interest of the new classes of oral CGRP inhibitors (gepants) and lasmitidan as a 5HT-1 agonist, there still is a significant unmet need for the safe and efficacious treatment of episodic migraine attacks.
PL37 has shown great efficacy in animal models of migraine. Based in these results, Pharmaleads will be starting a Phase 2a Proof of Concept study into the efficacy and safety of oral PL37 for the acute treatment of episodic migraine headache in 2020.
Replacement for injectable morphine in surgical, post surgical pain
Migraine is a frequent and debilitating painful disease, affecting more than 12% of the general population. NSAIDs and triptans are currently used for the acute treatment of migraine attacks. However, these treatments are not effective or well-tolerated in all patients. Despite the interest of the new classes of oral CGRP inhibitors (gepants) and lasmitidan as a 5HT-1 agonist, there still is a significant unmet need for the safe and efficacious treatment of episodic migraine attacks.
PL37 given orally has shown great efficacy in an animal model of migraine. Based on these results, Pharmaleads will be starting a Phase 2a Proof of Concept trial into the efficacy and safety of PL37 for the acute treatment of episodic migraine headache in 2020.
The efficacy of morphine without its side effects
Pharmaleads intends to pursue the development of PL37 in a Phase I study as a potential replacement for injectable morphine in surgical, post traumatic pain. The trial is expected to begin in 2021.
Current treatments include injectable and oral opioids which have significant side effects such as respiratory depression, constipation, nausea and sedation as well as dependence and tolerance issues.
The current global injectable opioids market is estimated to be worth $ 6 billion.
Pharmaleads is moreover constantly working to further its pipeline of DENKIs and new chemical families are still being designed, tested and patented.

